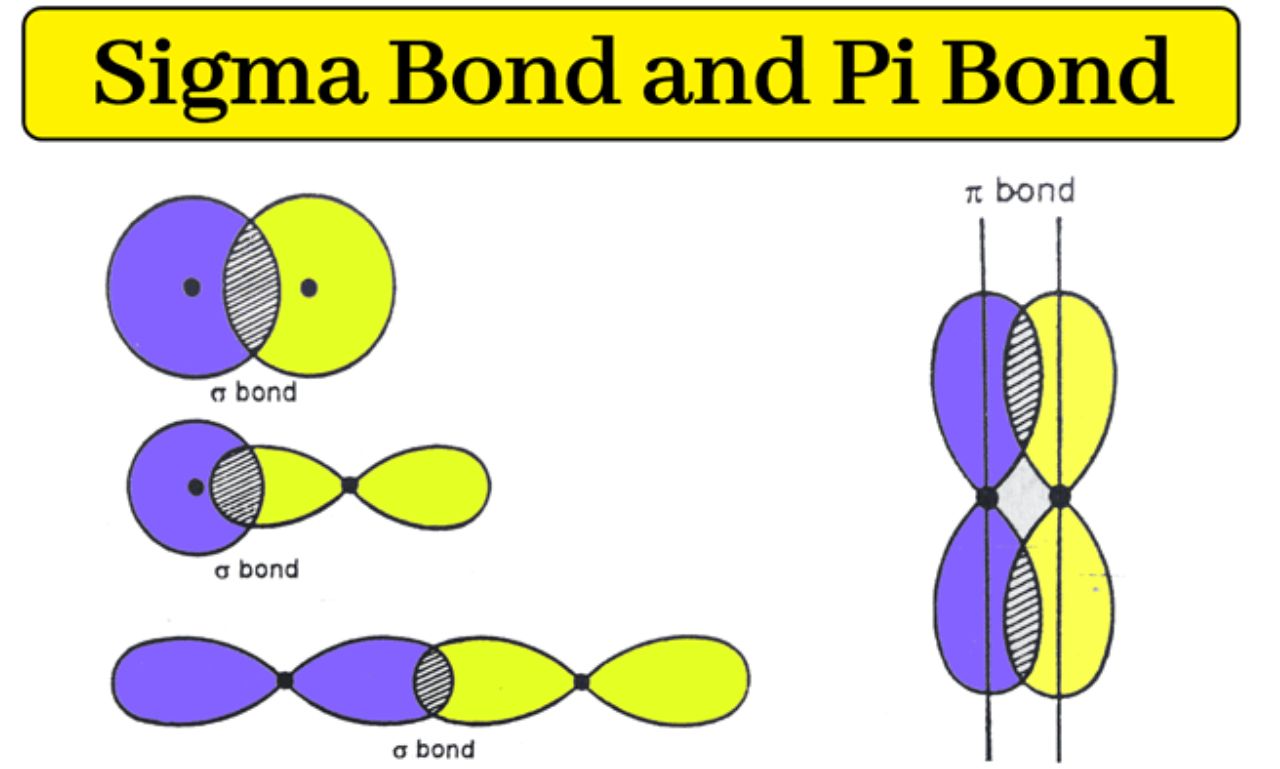
Orbital sigma (σ orbital) refers to a type of atomic or molecular orbital involved in forming sigma (σ) bonds, which are the strongest and most common chemical bonds between atoms. Sigma orbitals are created when atomic orbitals overlap directly along the axis connecting two atomic nuclei. These orbitals play a fundamental role in molecular structure and stability.
Understanding orbital sigma is important in chemistry because sigma bonds form the basic framework of most molecules.
What Is an Orbital Sigma?
An orb ital sigma is a molecular orbital formed by the head-on overlap of atomic orbitals. This overlap occurs along the line connecting the nuclei of two atoms, allowing electrons to be shared between atoms and creating a strong chemical bond.
Sigma orbitals are usually represented by the Greek letter σ (sigma) and are essential in covalent bonding.
Sigma orbitals can form from the overlap of different types of atomic orbitals, including:
-
s orbitals with s orbitals (s–s overlap)
-
s orbitals with p orbitals (s–p overlap)
-
p orbitals with p orbitals (p–p overlap)
Characteristics of Sigma Orbitals
Sigma orbitals have several unique characteristics that distinguish them from other orbitals.
1. Direct Overlap
Sigma orbitals form through direct or head-on overlap of atomic orbitals. This type of overlap creates a strong bond because electrons are concentrated between the nuclei.
2. Cylindrical Symmetry
Sigma orbitals have cylindrical symmetry around the bond axis. This means the electron density is evenly distributed around the line connecting the atoms.
3. Strong Bonds
Sigma bonds formed by sigma orbitals are generally stronger than other types of bonds because of effective orbital overlap.
4. Free Rotation
Molecules connected by a single sigma bond can usually rotate freely around the bond axis without breaking the bond.
Sigma Orbitals vs Pi Orbitals
Sigma orb itals are often compared with pi (π) orbitals, another important type of molecular orb ital.
| Feature | Sigma Orbital (σ) | Pi Orbital (π) |
|---|---|---|
| Overlap Type | Head-on overlap | Side-by-side overlap |
| Bond Strength | Stronger | Weaker |
| Rotation | Free rotation possible | Rotation restricted |
| Electron Density | Along bond axis | Above and below axis |
Sigma orb itals form the first bond between two atoms, while pi orb itals form additional bonds.
Examples of Sigma Orbitals
Sigma orbitals appear in many common molecules.
Hydrogen Molecule (H₂)
Two hydrogen atoms form a sigma bond through s–s orb ital overlap.
Methane (CH₄)
Carbon forms four sigma bonds with hydrogen atoms using hybrid orb itals.
Ethane (C₂H₆)
Carbon atoms are connected by a single sig ma bond.
These examples demonstrate how sig ma orbi tals create stable molecular structures.
Importance of Orbital Sigma
Sigma orbitals are essential in chemistry for several reasons.
Molecular Stability
Sigma bonds form the backbone of molecules and provide structural strength.
Chemical Reactions
Many reactions involve breaking or forming sigma bonds.
Molecular Geometry
Sigma orbi tals influence the shape and arrangement of molecules.
Basic Chemical Bonding
Every single covalent bond is a sigma bond.
Applications of Sigma Orbital Theory
Understanding orb ital sig ma helps in:
-
Predicting molecular shapes
-
Studying chemical reactions
-
Designing new materials
-
Understanding organic chemistry
-
Learning molecular bonding
Sigma orbital theory is a key part of modern chemistry education.
Conclusion on Orbital Sigma
Orbital sig ma refers to the molecular orbital responsible for forming sigma bonds through direct orbital overlap. These orbitals create strong and stable chemical bonds that form the foundation of molecular structures.
Learning about sigma orbitals helps explain how atoms bond together and why molecules have specific shapes and properties